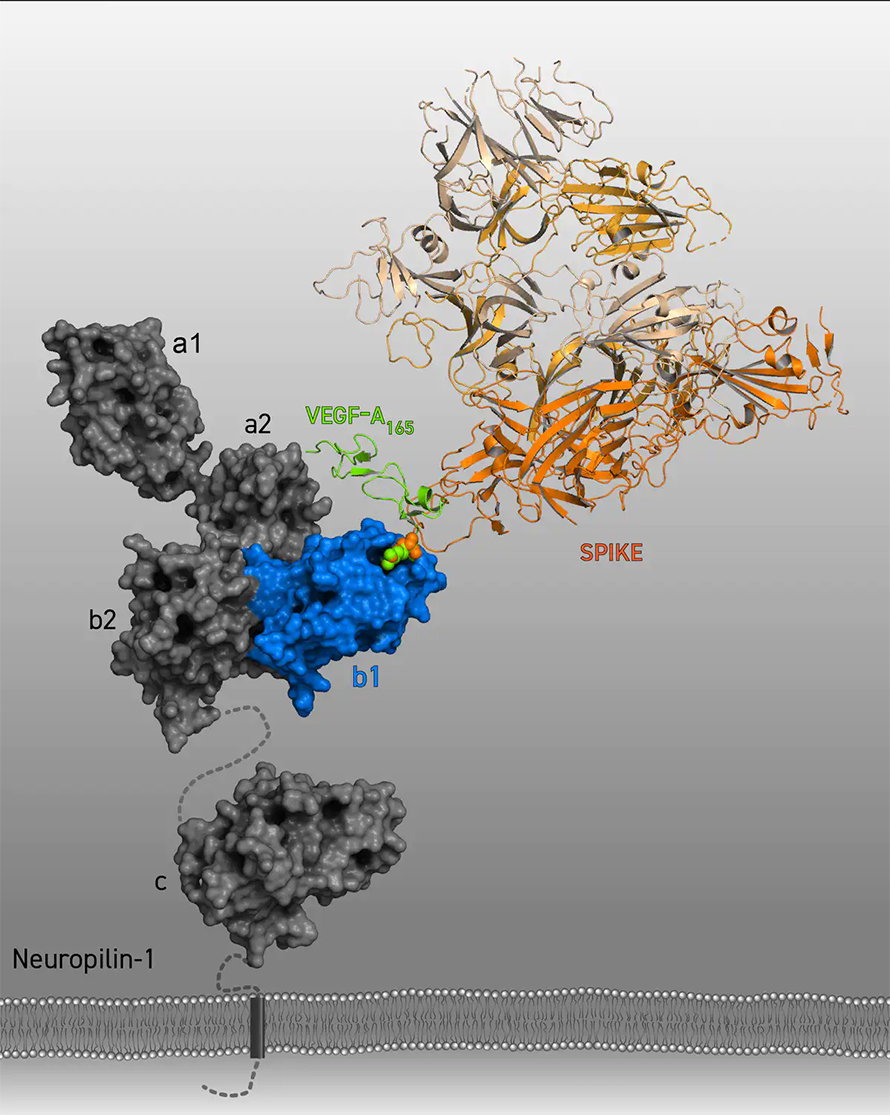
This finding likely reflects an underlying genetic susceptibility among the infected that absolutely needs to be defined and taken into account, particularly if HERV-W is going to be used as a general marker for disease severity, or as a therapeutic target for a humanized monoclonal antibody therapy, as is now envisioned. A few of these observations were initially pointed out to me by an anonymous researcher on social media operating with the moniker "Underground courtlady."Ī key finding in these studies is that not all COVID patients had significant HERV-W ENV activation only 20 or 30 percent of them did. Some vaccine manufacturers have eliminated the furin cleavage site from their mRNA construct in order to reduce potential residual fusion of a 2-PP stabilized construct. 
However, more research is needed to fully characterize the fusogenic potential of stabilized spike proteins. One way this has been done is to stabilize specific conformers through the addition of two strategic prolines to the code. mRNA vaccines therefore have slight modifications that simultaneously make the mRNA less immunogenic, and the spike protein it encodes more immunogenic. Native spike proteins tend to prematurely refold into a post-fusion conformation, which compromises immunogenic properties and prefusion trimer yields. Curiously, only the RNAs for HERV-W resulted in subsequent ENV protein expression. They found immediate and significant upregulation of the RNAs for the ENV protein from both HERV-W and HERV-K. Researchers added a recombinant trimeric spike protein without stabilizing mutations to cultured peripheral blood mononuclear cells (PBMCs) from SARS-CoV-2 patients. Now, the real culprit in HERV-W activation has been discovered. However, the exact mechanisms involved were not clear. Researchers had previously observed a correlation in the expression of HERV-W ENV protein in T lymphocytes with severe respiratory distress in SARS-CoV-2 patients. Apparently, the shape of the letter W somehow reminded the naming committee of the shape of the ring structure of atoms in the side chain of tryptophan. HERV-W is named for the fact that many retroviruses in the group use a tryptophan tRNA in the primer binding site. The authors provide solid evidence that the SARS-CoV-2 spike protein activates the envelope (ENV) protein encoded by HERV-W in blood cells, which is in turn directly responsible for many pathological features of the disease. On Tuesday, a real zinger was dropped onto the medRxiv preprint server that could potentially explain many of the commonly observed pathogenic features of SARS-CoV-2. 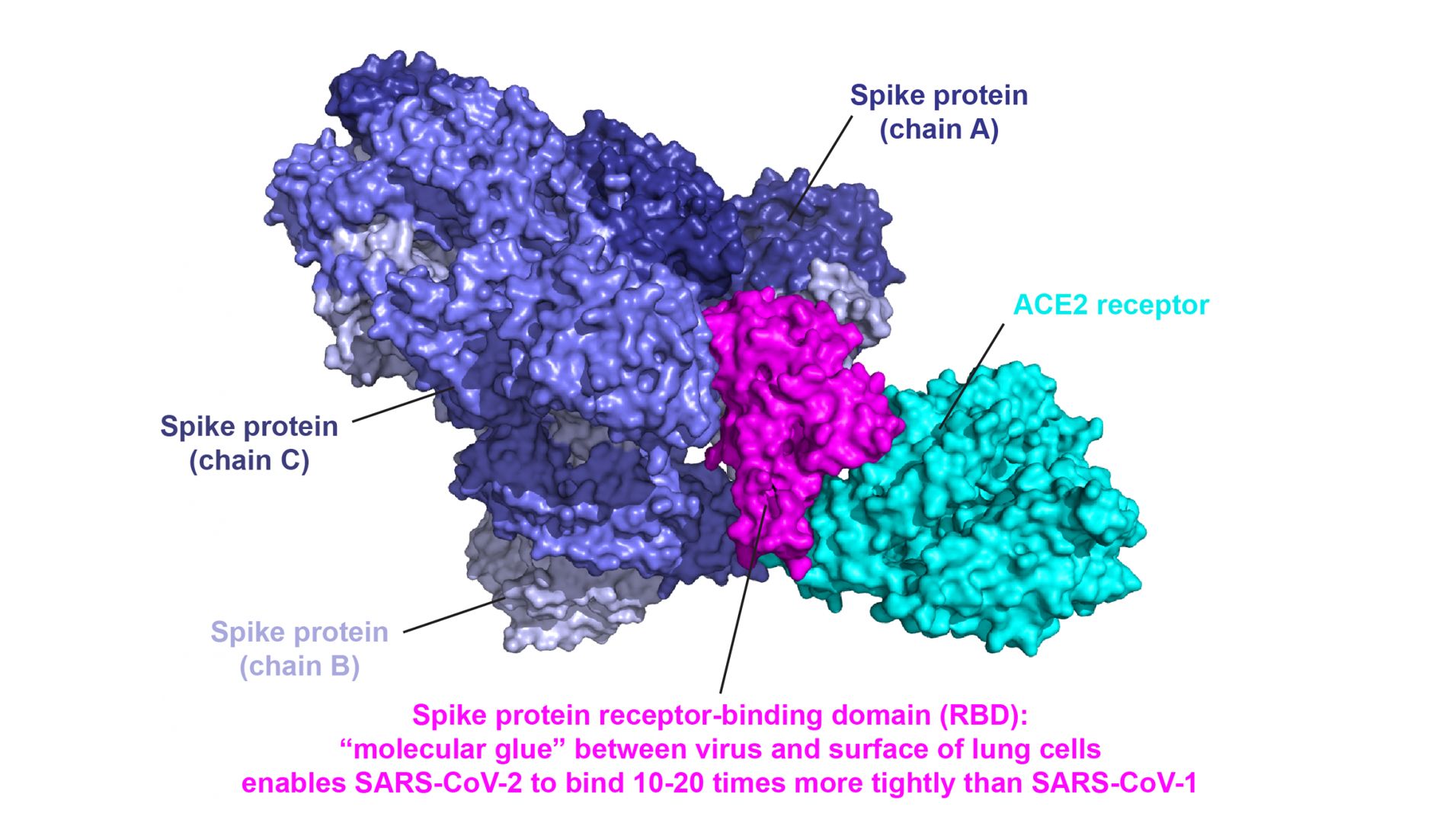
Retrotransposons derive from human endogenous retrovirus (HERVs) but typically have lost their signature long terminal repeat sequences at the beginning and ends of their genes. Last week, we reviewed the activation of one particular kind of transposable element, the Line-1 retrotransposons, in an ever-expanding host of neurodegenerative conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
